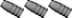Here is a simple test if you have metal that you don't know if it is zinc, or contains zinc, or if it is a good lead alloy.
First, tap it. If it rings, it is not pure lead.
Here's another way with a simple chemical you may already have at your house, or store.
This is a simple toilet cleaner called "The Works". According to the MSDS-
http://www.instructables.com/files/o...F8GN4CZIND.pdf
-it contains between 15% and 25% hydrochloric acid by weight.
One of these two ingots is nearly pure zinc. The other is an ingot of smelted range lead.
Can you tell which one is which by looking?
Simply squirt some acid on both ingots and observe the reaction.
The lead ingot hardly foams at all.
The zinc ingot foams a lot. In fact it foamed so much the acid solution was pushed off of the ingot and onto the concrete before I could snap the picture. The yellow color of the foam is a reaction with the concrete surface beneath the ingots.
Be aware- the reaction between zinc or lead + hydrochloric acid produces zinc or lead chloride and hydrogen gas, which is very flammable! So no open flames near the testing, ok?
While Lead will produce a slight amount of fizzing (lead will react with HCl) Zinc is much more reactive.
Hope this helps!

|
   
   
|


|


 Reply With Quote
Reply With Quote